Post updated 2014 January 2nd
DNA methylation is a biochemical process and epigenetic modification, whereby a methyl group is added to the cytosine nucleotide (and also adenine) to form 5-methylcytosine. DNA methylation at the 5' position of cytosine has the specific effect of reducing gene transcription and typically occurs in CpG sites, which are regions of DNA where a cytosine occurs next to a guanine (the p stands for the phosphate link that links them together). Most CpGs are methylated in mammals, however, unmethylated CpGs are often grouped in clusters called CpG islands, which are present in the 5' regulatory regions of many genes.
One way of profiling methylation patterns in DNA is via the use of bisulphite treatment of DNA following by sequencing, which is known as bisulphite sequencing. The treatment of DNA with bisulphite converts cytosine residues to uracil, but leaves 5-methylcytosine residues unaffected. Therefore after sequencing, cytosine residues represent methylated cytosines in the genome. One variant of bisulphite sequencing, is reduced representation bisulphite sequencing (RRBS), which was developed as a cost-efficient method to profile areas of the genome that have a high CpG content. Genomic DNA is digested using the restriction endonuclease MspI, which recognises the sequence 5'-CCGG-3'. MspI is actually part of a isoschizomer pair with HpaII, which are restriction enzymes that are specific to the same recognition sequence. However MspI can recognise methylated cytosines, whereby HpaII cannot.
MspI site MspI site
5'-...CCGGNNNNNNNNNNNNNNNNNNNNNCCGG...-3'
3'-...GGCCNNNNNNNNNNNNNNNNNNNNNGGCC...-5'
MspI digestion
5'- CGGNNNNNNNNNNNNNNNNNNNNNC -3'
3'- CNNNNNNNNNNNNNNNNNNNNNGGC -5'
The ENCODE project generated a lot of RRBS data for different samples and two whole genome bisulphite sequencing (WGBS) datasets on H1hESC and GM12878. This thread on Nature aggregated the DNA methylation papers published as part of ENCODE.
The ENCODE reduced-representation bisulphite sequencing (RRBS) methylation data can be found at this URL:
http://hgdownload.cse.ucsc.edu/goldenPath/hg19/encodeDCC/wgEncodeHaibMethylRrbs/
I will use the RRBS data on the K562 cell line and the H1hESC cell line.
#download the files wget http://hgdownload.cse.ucsc.edu/goldenPath/hg19/encodeDCC/wgEncodeHaibMethylRrbs/wgEncodeHaibMethylRrbsK562HaibSitesRep1.bed.gz wget http://hgdownload.cse.ucsc.edu/goldenPath/hg19/encodeDCC/wgEncodeHaibMethylRrbs/wgEncodeHaibMethylRrbsK562HaibSitesRep2.bed.gz
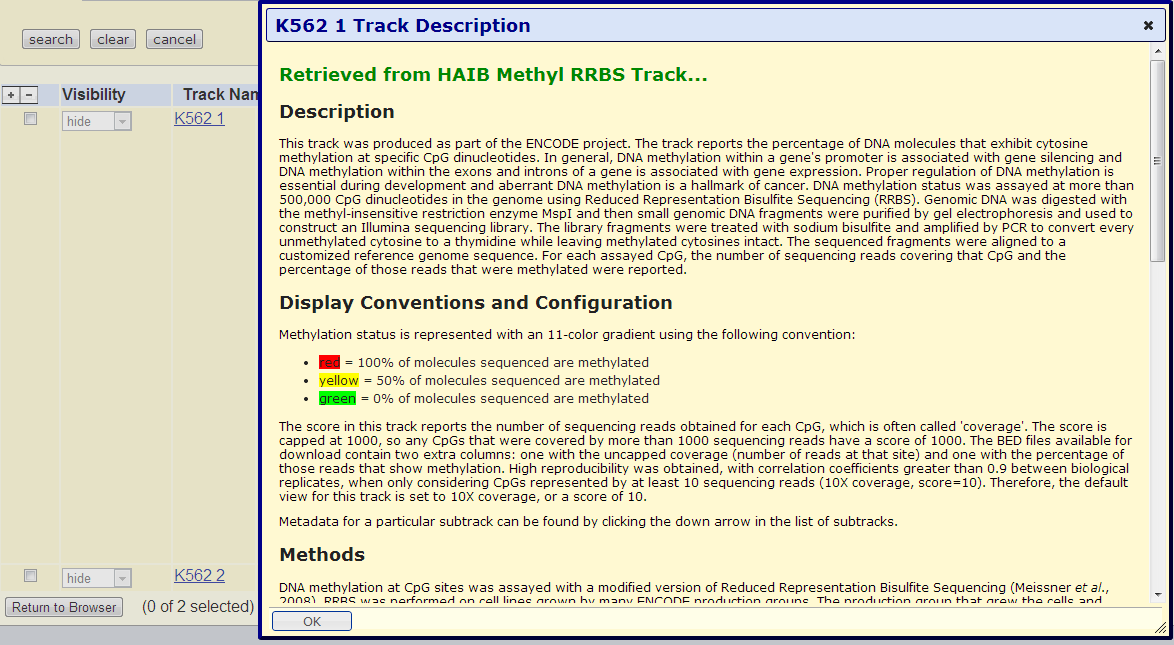
For more information on these tracks, use the UCSC Genome Browser search page, then click on "Track search" under "Search Human Genome GRCh37/hg19" and in the "Track Name" box type "Methyl-RRBS" and for "Cell, tissue or DNA sample", select K562. You should get two results; clicking on "K562 1" will give you the full description of these files.
From the "Display Conventions and Configuration" section it reads:
The BED files available for download contain two extra columns: one with the uncapped coverage (number of reads at that site) and one with the percentage of those reads that show methylation. High reproducibility was obtained, with correlation coefficients greater than 0.9 between biological replicates, when only considering CpGs represented by at least 10 sequencing reads (10X coverage, score=10). Therefore, the default view for this track is set to 10X coverage, or a score of 10.
If we look into the file, we can see the typical BED columns (1-9) and the two additional columns (10-11):
chr start end name score strand tStart tEnd rgb count percentMeth chr1 10785 10786 K562_Rep3_RRBS 4 + 10785 10786 255,0,0 4 100 chr1 88704 88705 K562_Rep3_RRBS 3 + 88704 88705 255,155,0 3 67 chr1 137976 137977 K562_Rep3_RRBS 35 + 137976 137977 255,55,0 35 94
So at position chr1:10785-10786, all 4 reads were methylated and at position chr1:88704-88705, 2 of the 3 reads were methylated. Let's calculate some simple statistics:
#how many profiled positions in the first replicate
zcat wgEncodeHaibMethylRrbsK562HaibSitesRep1.bed.gz | grep -v "^track" | wc -l
1275818
#how many profiled positions with at least 10 sequencing reads?
zcat wgEncodeHaibMethylRrbsK562HaibSitesRep1.bed.gz | grep -v "^track" | awk '$5>9 {print}' | wc -l
808212
#what is the distribution of read counts at
#each profiled position?
zcat wgEncodeHaibMethylRrbsK562HaibSitesRep1.bed.gz | grep -v "^track" | cut -f10 | stats
Total lines: 1275818
Sum of lines: 45676252
Ari. Mean: 35.8015422262423
Geo. Mean: 13.4023455040408
Median: 21
Mode: 1 (N=208629)
Anti-Mode: 382 (N=1)
Minimum: 1
Maximum: 32552
Variance: 34252.6155229694
StdDev: 185.074621498923
#what is the distribution of methylation percentage?
zcat wgEncodeHaibMethylRrbsK562HaibSitesRep1.bed.gz | grep -v "^track" | cut -f11 | stats
Total lines: 1275818
Sum of lines: 37255124
Ari. Mean: 29.2009706713654
Geo. Mean: undef (zero found in data)
Median: 4
Mode: 0 (N=554420)
Anti-Mode: 34 (N=1418)
Minimum: 0
Maximum: 100
Variance: 1514.74526361253
StdDev: 38.9197284627287
So for the first replicate of the K562 RRBS dataset (wgEncodeHaibMethylRrbsK562HaibSitesRep1.bed.gz), 1,275,818 sites were profiled (808,212 with 10 reads or more) and the median distribution of reads sequenced was 21 and the median methylation percentage was 4.
Let's compare this to human embryonic stem cells:
wget http://hgdownload.cse.ucsc.edu/goldenPath/hg19/encodeDCC/wgEncodeHaibMethylRrbs/wgEncodeHaibMethylRrbsH1hescHaibSitesRep1.bed.gz
#profiled sites
zcat wgEncodeHaibMethylRrbsH1hescHaibSitesRep1.bed.gz | grep -v "^track" | wc -l
1288079
#profiled sites with >=10 reads
zcat wgEncodeHaibMethylRrbsH1hescHaibSitesRep1.bed.gz | grep -v "^track" | awk '$5>9 {print}' | wc -l
713920
#distribution of read counts
zcat wgEncodeHaibMethylRrbsH1hescHaibSitesRep1.bed.gz | grep -v "^track" | cut -f10 | stats
Total lines: 1288079
Sum of lines: 28218867
Ari. Mean: 21.9077145112994
Geo. Mean: 9.53485643942492
Median: 12
Mode: 1 (N=204533)
Anti-Mode: 247 (N=1)
Minimum: 1
Maximum: 18154
Variance: 11753.75583868
StdDev: 108.414739951171
#distribution of percentage methylation
zcat wgEncodeHaibMethylRrbsH1hescHaibSitesRep1.bed.gz | grep -v "^track" | cut -f11 | stats
Total lines: 1288079
Sum of lines: 42226713
Ari. Mean: 32.782704321707
Geo. Mean: undef (zero found in data)
Median: 2
Mode: 0 (N=601926)
Anti-Mode: 51 (N=91)
Minimum: 0
Maximum: 100
Variance: 1873.49808066919
StdDev: 43.2839240442591
Statistics via R
Calculating some statistics and making plots using R:
data <- read.table(gzfile("wgEncodeHaibMethylRrbsH1hescHaibSitesRep1.bed.gz"),
header=F,
skip=1)
head(data)
# V1 V2 V3 V4 V5 V6 V7 V8 V9 V10 V11
#1 chr1 1000170 1000171 SL716_RRBS 41 + 1000170 1000171 255,105,0 41 83
#2 chr1 1000190 1000191 SL716_RRBS 41 + 1000190 1000191 255,105,0 41 80
#3 chr1 1000191 1000192 SL716_RRBS 24 - 1000191 1000192 255,105,0 24 75
#4 chr1 1000198 1000199 SL716_RRBS 41 + 1000198 1000199 255,0,0 41 98
#5 chr1 1000199 1000200 SL716_RRBS 24 - 1000199 1000200 255,55,0 24 92
#6 chr1 1000206 1000207 SL716_RRBS 24 - 1000206 1000207 255,0,0 24 96
#number of reads mapped
sum(data$V10)
#[1] 28218867
#density of methylation percent
plot(density(data$V11))
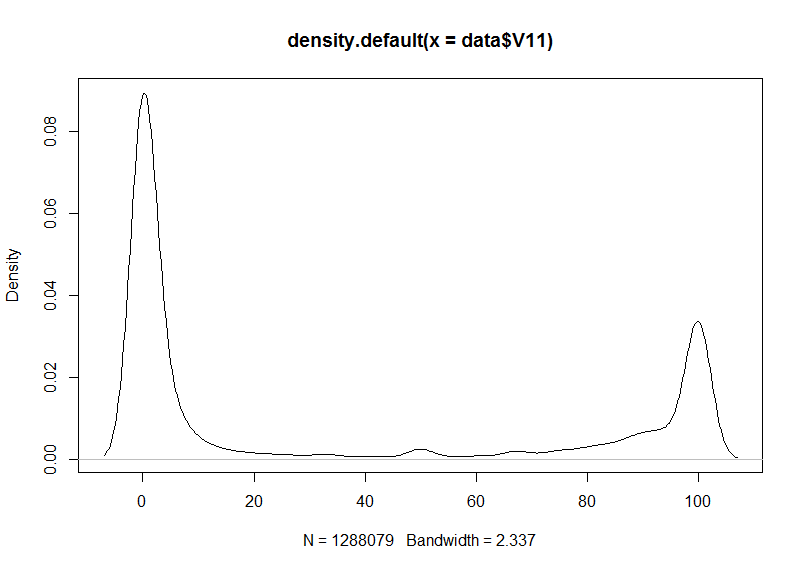 Bimodal distribution of methylation percentage in the H1ESC RRBS dataset.
Bimodal distribution of methylation percentage in the H1ESC RRBS dataset.
And for the K562 cell line:
data <- read.table(gzfile("wgEncodeHaibMethylRrbsK562HaibSitesRep1.bed.gz"),
header=F,
skip=1)
#number of sites assayed
sum(data$V10)
[1] 45676252
plot(density(data$V11))
 Bimodal distribution of methylation percentage in the K562 RRBS dataset.
Bimodal distribution of methylation percentage in the K562 RRBS dataset.
Distribution of mapped reads at each position:
h1 <- read.table(gzfile("wgEncodeHaibMethylRrbsH1hescHaibSitesRep1.bed.gz"),
header=F,
skip=1)
k562 <- read.table(gzfile("wgEncodeHaibMethylRrbsK562HaibSitesRep1.bed.gz"),
header=F,
skip=1)
#number of sites profiled
dim(h1)
[1] 1288079 11
dim(k562)
[1] 1275818 11
#distribution of read counts
quantile(h1$V10, probs=seq(0,1,0.1))
0% 10% 20% 30% 40% 50% 60% 70% 80% 90% 100%
1 1 2 4 8 12 18 25 35 53 18154
quantile(k562$V10, probs=seq(0,1,0.1))
0% 10% 20% 30% 40% 50% 60% 70% 80% 90% 100%
1 1 2 5 12 21 30 41 56 82 32552
Methylation status of CpG islands
Obtain CpG islands:
echo 'SELECT * FROM cpgIslandExt' |
mysql -B --user=genome --host=genome-mysql.cse.ucsc.edu hg19 |
grep -v "^bin" | awk 'OFS="\t" {print $2, $3, $4}' > hg19_cpg_island.bed
md5sum hg19_cpg_island.bed
2efe7d09742fbd7d5668dcd0c8d15db3 hg19_cpg_island.bed
Intersect with assayed sites in K562 and H1hesc:
intersectBed -a wgEncodeHaibMethylRrbsK562HaibSitesRep1.bed.gz -b hg19_cpg_island.bed -u | gzip > k562_rep1_cpg_island.bed.gz #number of sites overlapping zcat k562_rep1_cpg_island.bed.gz | wc -l 759650 #how many total zcat wgEncodeHaibMethylRrbsK562HaibSitesRep1.bed.gz | wc -l 1275819 #percent of total overlapping CpG islands bc -l<<<759650*100/1275819 59.54214508484353971840 #distribution of methylation percentage zcat k562_rep1_cpg_island.bed.gz | cut -f11 | stats Total lines: 759650 Sum of lines: 16809084 Ari. Mean: 22.1274060422563 Geo. Mean: undef (zero found in data) Median: 1 Mode: 0 (N=372779) Anti-Mode: 49 (N=862) Minimum: 0 Maximum: 100 Variance: 1207.24553825536 StdDev: 34.7454391000511 #how with h1hesc intersectBed -a wgEncodeHaibMethylRrbsH1hescHaibSitesRep1.bed.gz -b hg19_cpg_island.bed -u | gzip > h1hesc_rep1_cpg_island.bed.gz zcat h1hesc_rep1_cpg_island.bed.gz | wc -l 795124 zcat h1hesc_rep1_cpg_island.bed.gz | cut -f11 | stats Total lines: 795124 Sum of lines: 8410792 Ari. Mean: 10.5779626825501 Geo. Mean: undef (zero found in data) Median: 0 Mode: 0 (N=510970) Anti-Mode: 66 (N=32) Minimum: 0 Maximum: 100 Variance: 721.801261304316 StdDev: 26.8663592863699
Methylation of regions upstream of RefSeq models
The upstream1000.fa.gz file contains sequences 1000 bases upstream of annotated transcription starts of RefSeq genes with annotated 5' UTRs.
wget http://hgdownload-test.cse.ucsc.edu/goldenPath/hg19/bigZips/upstream1000.fa.gz zcat upstream1000.fa.gz | grep "^>" | cut -f2 -d' ' | sed 's/[:-]/\t/g' | gzip > upstream1000.bed.gz intersectBed -a wgEncodeHaibMethylRrbsH1hescHaibSitesRep1.bed.gz -b upstream1000.bed.gz -u | gzip > h1hesc_rep1_upstream1000.bed.gz intersectBed -a wgEncodeHaibMethylRrbsK562HaibSitesRep1.bed.gz -b upstream1000.bed.gz -u | gzip > k562_rep1_upstream1000.bed.gz zcat h1hesc_rep1_upstream1000.bed.gz | wc -l 237219 zcat k562_rep1_upstream1000.bed.gz | wc -l 225752
Using the R package methylKit
Altuna has developed a R package called methylKit, which provides analytical tools for DNA methylation data from high-throughput bisulfite sequencing. He has written a post on using ENCODE methylation data in R, which I will follow here. The track line definition in the BED files causes a problem for the package, so we must first remove them:
zcat wgEncodeHaibMethylRrbsH1hescHaibSitesRep1.bed.gz | grep -v "^track" | gzip > h1hesc_rep1.bed.gz zcat wgEncodeHaibMethylRrbsK562HaibSitesRep1.bed.gz | grep -v "^track" | gzip > k562_rep1.bed.gz
Now the analysis using R:
source("http://methylkit.googlecode.com/files/install.methylKit.R")
# installing the package with the dependencies
install.methylKit(ver="0.9.2",dependencies=TRUE)
library(methylKit)
file <- list("h1hesc_rep1.bed.gz",
"k562_rep1.bed.gz")
obj <- read(file,
sample.id = list("H1hesc", "K562"),
treatment = c(1,0),
assembly = "hg19",
header = FALSE,
context = "CpG",
resolution = "base",
pipeline = list(fraction = FALSE,
chr.col = 1,
start.col = 3,
end.col = 3,
coverage.col = 5,
strand.col = 6,
freqC.col = 11
)
)
obj
methylRawList object with 2 methylRaw objects
methylRaw object with 1288079 rows
--------------
chr start end strand coverage numCs numTs
1 chr1 1000171 1000171 + 41 34 7
2 chr1 1000191 1000191 + 41 33 8
3 chr1 1000192 1000192 - 24 18 6
4 chr1 1000199 1000199 + 41 40 1
5 chr1 1000200 1000200 - 24 22 2
6 chr1 1000207 1000207 - 24 23 1
--------------
sample.id: H1hesc
assembly: hg19
context: CpG
resolution: base
methylRaw object with 1275818 rows
--------------
chr start end strand coverage numCs numTs
1 chr1 1000171 1000171 + 46 16 30
2 chr1 1000191 1000191 + 46 7 39
3 chr1 1000192 1000192 - 53 5 48
4 chr1 1000199 1000199 + 46 9 37
5 chr1 1000200 1000200 - 53 8 45
6 chr1 1000207 1000207 - 53 14 39
--------------
sample.id: K562
assembly: hg19
context: CpG
resolution: base
treament: 1 0
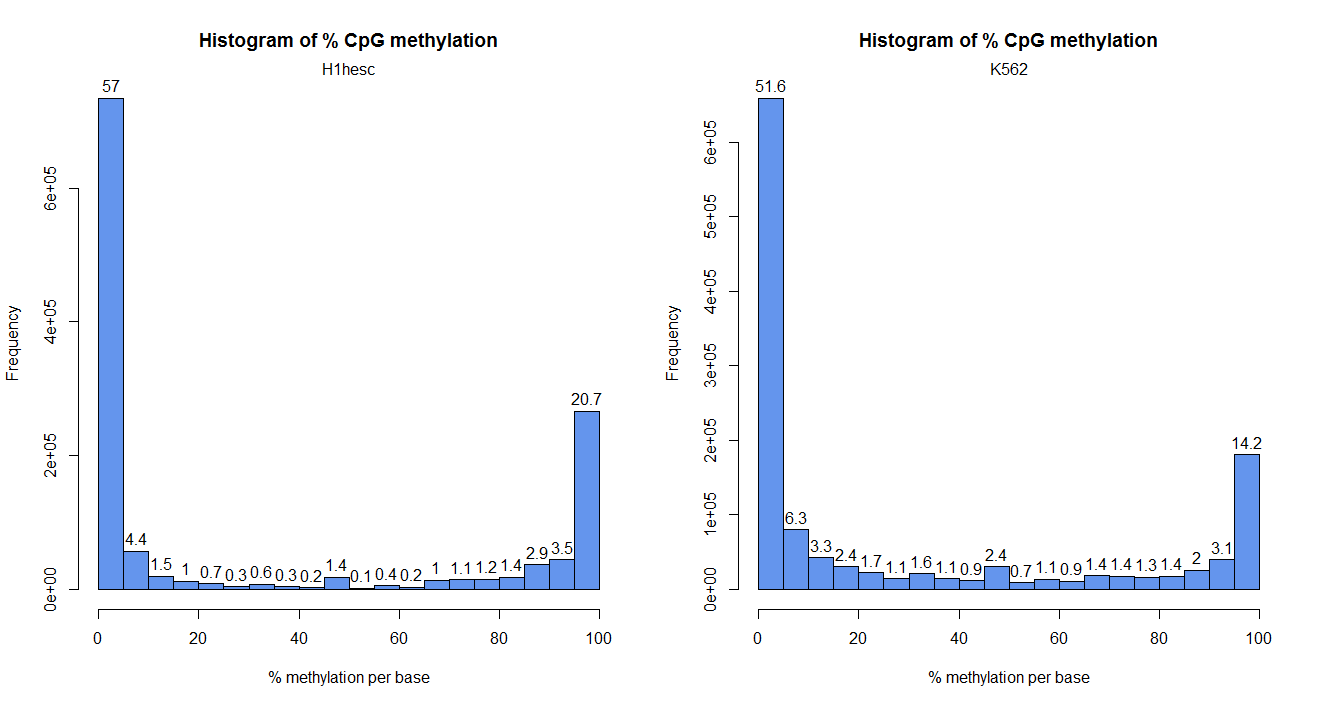
par(mfrow=c(1,2))
getMethylationStats(obj[[1]], plot = TRUE)
getMethylationStats(obj[[2]], plot = TRUE)
par(mfrow=c(1,1))
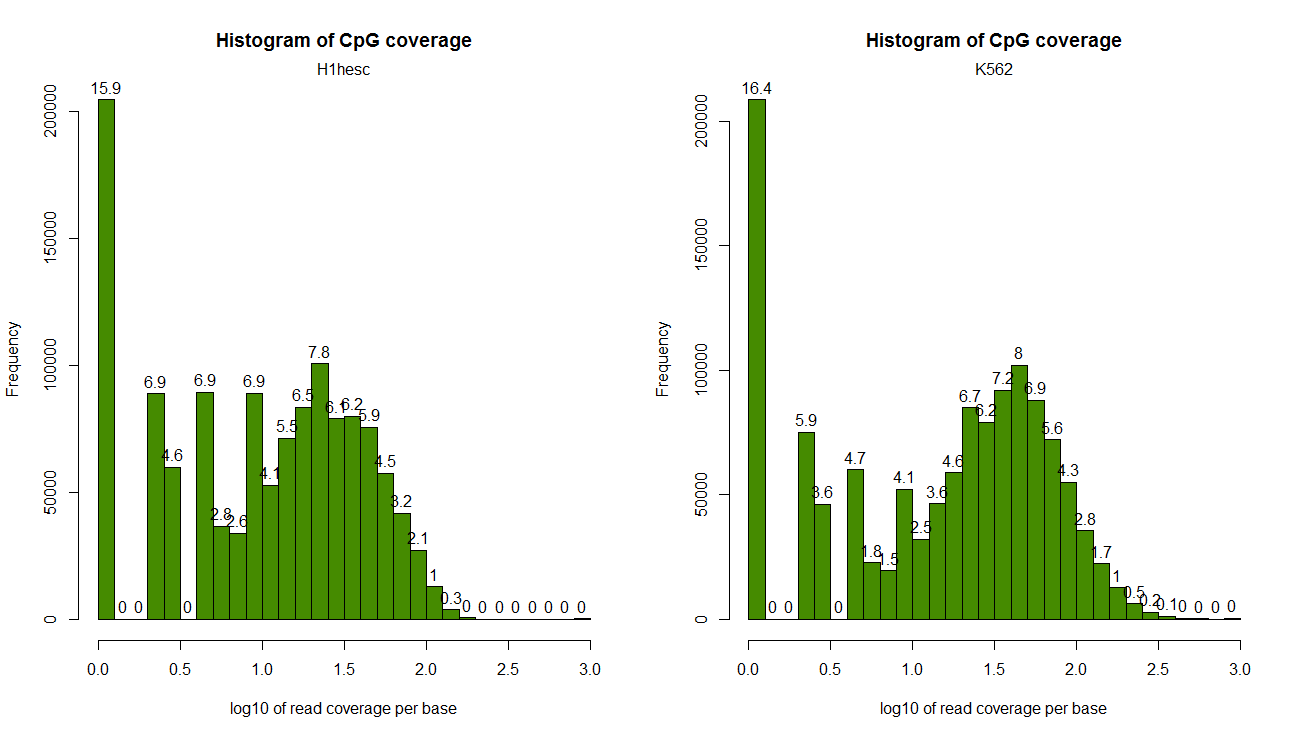
Read coverage per base:
par(mfrow=c(1,2)) getCoverageStats(obj[[1]],plot=T,both.strands=F) getCoverageStats(obj[[2]],plot=T,both.strands=F)
Finding differentially methylated bases or regions
meth <- unite(obj, destrand=FALSE)
head(meth)
methylBase object with 6 rows
--------------
chr start end strand coverage1 numCs1 numTs1 coverage2 numCs2 numTs2
1 chr1 88705 88705 + 3 2 1 3 2 1
2 chr1 137977 137977 + 2 2 0 35 33 2
3 chr1 137986 137986 + 2 2 0 35 28 7
4 chr1 713376 713376 + 11 9 2 35 9 26
5 chr1 713388 713388 + 11 10 1 35 6 29
6 chr1 713400 713400 + 11 10 1 35 2 33
--------------
sample.ids: H1hesc K562
destranded FALSE
assembly: hg19
context: CpG
treament: 1 0
resolution: base
myDiff <- calculateDiffMeth(meth, num.cores=16)
diffMethPerChr(myDiff, plot=F, qvalue.cutoff=0.01, meth.cutoff=25)
$diffMeth.per.chr
chr number.of.hypomethylated percentage.of.hypomethylated
1 chr1 9919 10.308668
12 chr2 8133 11.563069
16 chr3 4078 9.178483
17 chr4 3977 10.153956
18 chr5 5224 11.946032
19 chr6 4645 10.341296
20 chr7 5699 9.715309
21 chr8 4600 10.528243
22 chr9 4173 8.453015
2 chr10 5270 10.879214
3 chr11 5700 9.891368
4 chr12 3843 8.331165
5 chr13 2201 9.940385
6 chr14 3138 9.737177
7 chr15 4044 12.907344
8 chr16 3413 5.869606
9 chr17 7073 10.330826
10 chr18 2787 12.794381
11 chr19 5340 6.728152
13 chr20 3580 10.135613
14 chr21 1115 6.548039
15 chr22 3259 9.583603
23 chrM 0 0.000000
24 chrX 2326 9.642650
25 chrY 0 0.000000
number.of.hypermethylated percentage.of.hypermethylated
1 9994 10.386614
12 7120 10.122839
16 3163 7.119064
17 4216 10.764164
18 4835 11.056483
19 4265 9.495291
20 6763 11.529151
21 4766 10.908175
22 5138 10.407762
2 5970 12.324271
3 5645 9.795925
4 3957 8.578304
5 2581 11.656580
6 2816 8.738015
7 2581 8.237847
8 6072 10.442499
9 5023 7.336595
10 2582 11.853280
11 5474 6.896986
13 3990 11.296396
14 2857 16.778248
15 3617 10.636358
23 0 0.000000
24 1617 6.703424
25 107 36.518771
$diffMeth.all
percentage.of.hypermethylated number.of.hypermethylated
1 9.855018 105149
percentage.of.hypomethylated number.of.hypomethylated
1 9.703934 103537
Warning message:
In eval(expr, envir, enclos) : NAs introduced by coercion
#storing the results as a data.frame
data <- data.frame(chr=myDiff$chr, start=myDiff$start, end=myDiff$end, strand=myDiff$strand, pvalue=myDiff$pvalue, qvalue=myDiff$qvalue, diff=myDiff$meth.diff)
head(data)
chr start end strand pvalue qvalue diff
1 chr1 88705 88705 + 1.000000e+00 4.668054e-01 0.000000
2 chr1 137977 137977 + 1.000000e+00 4.668054e-01 5.714286
3 chr1 137986 137986 + 1.000000e+00 4.668054e-01 20.000000
4 chr1 713376 713376 + 1.471833e-03 3.660600e-03 56.103896
5 chr1 713388 713388 + 1.833536e-05 6.080518e-05 73.766234
6 chr1 713400 713400 + 1.691055e-07 7.280300e-07 85.194805
head(data[order(data$qvalue),])
chr start end strand pvalue qvalue diff
414424 chr17 31149809 31149809 - 2.363853e-222 5.886729e-217 -68.00000
414433 chr17 31149838 31149838 - 2.363853e-222 5.886729e-217 -68.00000
1066882 chrY 13489947 13489947 - 6.486169e-209 1.076839e-203 82.95817
802144 chr4 191003226 191003226 + 9.833213e-201 1.224388e-195 72.99165
1066803 chrY 13470831 13470831 + 3.185868e-197 3.173521e-192 87.06726
1066751 chrY 13462178 13462178 + 1.783279e-195 1.268834e-190 66.03226
Checking chr17:31149808-31149809 in the original files:
zcat h1hesc_rep1.bed.gz | grep chr17 | grep 31149809 chr17 31149808 31149809 SL716_RRBS 1000 - 31149808 31149809 55,255,0 6108 14 zcat k562_rep1.bed.gz | grep chr17 | grep 31149809 chr17 31149808 31149809 K562_Rep3_RRBS 1000 - 31149808 31149809 255,105,0 10775 82
We see that this site has the maximum read coverage, and differentially methylated (14% versus 82%) between h1hESC and K562 (hypomethylated in h1hESC).
See also

This work is licensed under a Creative Commons
Attribution 4.0 International License.

be careful when saying “K562 bisulfite sequencing data” — recall that K562 was only assayed for RRBS, unlike GM12878 and H1, which underwent whole-genome bisulfite sequencing. The latter is more informative when comparing to DHS peaks, etc. since it is not biased towards Msp/Hpa cut sites, which tend to reside in CpG-rich regions.
(a lot of tissue-specific methylation (or lack thereof) occurs outside these regions)
Hi Tim,
Thank you for making an important point. I can now appreciate why most of the sites I found were residing in CpG-rich regions. I will update this post later on and compare RRBS with WGBS data.
Thanks again!
Dave
Hi, please correct me if I’m misunderstanding something about this, but I’m not sure this bit does what you say it does:
My understanding is that the Encode RRBS data files contain every site to which reads were mapped, not just the ones that are methylated. So counting up all the lines in the file tells you how many sites were assayed in total, not how many of them were methylated. See [table schema](http://encodeproject.org/cgi-bin/hgTables?db=hg19&hgta_group=regulation&hgta_track=wgEncodeHaibMethylRrbs&hgta_doSchema=describe+table+schema) — it contains a column called “percentMeth”, and in the data files, many of these values are 0 indicating completely unmethylated positions.
Hi Bonnie,
Thanks for the comment; you are completely correct!
I will amend the post accordingly.
Cheers,
Dave
Hi Dave,
I just came across this page while searching for DNA methylation data. It is very nice to see that you documented the ENCODE DNA methylation data and its analyses so well. I have downloaded the .bed.gz files from “http://hgdownload.cse.ucsc.edu/goldenPath/hg19/encodeDCC/wgEncodeHaibMethylRrbs/” but could not open it. I tried using gunzip and tar but faliled. Can u plz suggest me how to open/extract this extension?
Hi Subhadeep,
how did you download the files and how are you trying to open the files?
Cheers,
Dave
I downloaded the file from “http://hgdownload.cse.ucsc.edu/goldenPath/hg19/encodeDCC/wgEncodeHaibMethyl450/”
the file name is “wgEncodeHaibMethyl450Gm12878SitesRep1.bed.gz”
I tried to open using wget command. tried to open by “gunzip” and “tar -xzvf” and got the message “gunzip: wgEncodeHaibMethyl450Gm12878SitesRep1.bed.gz: not in gzip format”. :/
sorry! i Tried to open using gunzip command. downloaded using wget.
The gunzip command will extract the file. For example, if you ran:
The file would now become wgEncodeHaibMethyl450Gm12878SitesRep1.bed. You can view the contents of this file by running:
yeah I tried that but this msg appears:
“gunzip: wgEncodeHaibMethyl450Gm12878SitesRep1.bed.gz: not in gzip format”.
may be the files are corrupted.
Anyway, thanks a lot.
Hi Dave,
When talking about 10X coverage, should I use score no less than 10 or #reads of that site no less than 10? Most of the time, they have the same value, but there does exist some sites with different values. I am not sure whether it works for both WGBS and RRBS.
Thanks,
Sam
Hi Sam,
the score column should be the same as count column and as far as I can remember, the coverage is calculated by the number of reads. I can’t advise on what threshold you should use. In wgEncodeHaibMethylRrbsK562HaibSitesRep1.bed.gz, the median read count was 21.
Cheers,
Dave